Unser gemeinnütziger Verein „Waisenmedizin“ – Promoting Access to Care with Essential Medicine (PACEM) e. V.) hat fast 20 Jahre lang klinisch geforscht, um herauszufinden, wie die Heilung der Hautleishmaniose in der sog. Alten Welt effizienter, d. h. kosmetisch akzeptabler und preiswerter gemacht werden kann als die seit über 50 Jahren praktizierte äußert schmerzhafte Chemotherapie mit penta-valenten Antimonpräparaten. Die Leishmanien-Parasiten, welche die Hautläsionen auslösen, werden durch blutsaugende Sandfliegen in die menschliche Haut vor allem im Gesicht und wieder überwiegend bei Kleinkindern mit leicht zu durchstechender Haut übertragen, wo sie sich in den großen Abwehrzellen, den Hautmakrophagen, vermehren, deren Verteidigung sie widerstehen können und die dadurch gehindert werden, zur Wundheilung beizutragen.
Zwei Phase-II-Studien, die in internationalen Peer-Review-Zeitschriften (PLOS und BMC) veröffentlicht und vom National Institute for Health and Care Excellence (NICE) zitiert wurden, haben gezeigt, dass Hautleishmaniose in ihrer ulzerösen Form eine chronische Wundkrankheit ist. Sie ist keineswegs tödlich, außer in Ausnahmefällen einer Immunschwäche. Die Antimonchemotherapie beseitigt den Parasiten nicht vollständig aus dem menschlichen Körper, es gibt keine „Parasiten sterile“ Heilung. Vielmehr überleben verbleibende Parasiten in den Zellen unseres retikulo-endothelialen Systems einschließlich der Makrophagen, einem Zellsystem, das für unsere unspezifische Immunabwehr so wichtig ist. Auch wenn keine Wundbehandlung durchgeführt wird, steht am Ende von Monaten oder Jahren eine Spontanheilung, jedoch mit einem mehr oder weniger entstellenden, lebenslangen unästhetischen Ergebnis. Diese Narben stigmatisieren Mädchen und schließen Frauen aus sehr konservativen Gesellschaften in Ländern wie Afghanistan, Pakistan und einigen arabischen Ländern aus.
Die Empfehlung von KITAB EÇ ÇIH’HA in dem Buch der guten Gesundheit, „Le Livre de la Bonne Santé“, das 1922 in Algerien veröffentlicht wurde, ist mit dem Aufkommen der Antimon-Chemotherapie vor über 50 Jahren für offene Läsionen der Hautleishmaniose der Alten Welt vergessen worden. Wir haben uns bemüht, diese Empfehlungen unter Berücksichtigung der von der WHO empfohlenen Handhygiene einzuhalten. Diese Methode ist in der Lage, komplexe Läsionen an Körperstellen, die einer topischen Antimonbehandlung unzugänglich sind, in kurzer Zeit von wenigen Wochen zu heilen. Auf eine wirtschaftlichere und weniger schmerzhafte Weise führt dies zu besseren ästhetischen Ergebnissen und kürzeren Heilungszeiten.
Mit unseren Pilotergebnissen im L. major Leishmania Endemie Gebiet von Bou Saada und M’Sila in Algerien haben wir die vorläufige Sonderzulassung nach § 11 MPG (Medizinproduktegesetz), die es nur in EU Ländern wie Deutschland gibt, für unsere LeiClean-Behandlung beantragt, wir haben die Sonderzulassung erhalten und inzwischen zum zweiten Mal bis September 2020 verlängert bekommen. Diese Sonderzulassung gilt vorübergehend, sie ersetzt keinesfalls die CE Zertifizierung durch den TÜV (Technischer Überwachungsverein).
Auch wenn der verantwortliche Hersteller, wie in unserem Fall, ein gemeinnütziger Verein ist, ist er verpflichtet, die Kosten zu tragen, und diese Kosten betragen rund 60.000 €. Die CE-Zertifizierung ist notwendig, damit die Leishmania endemischen Länder die für die LeiClean-Behandlung notwendigen Materialien im eigenen Land für ihre Leishmania Patienten herstellen können, nachdem sie eine lokale Validierung der CE-Zertifizierung erreicht haben.
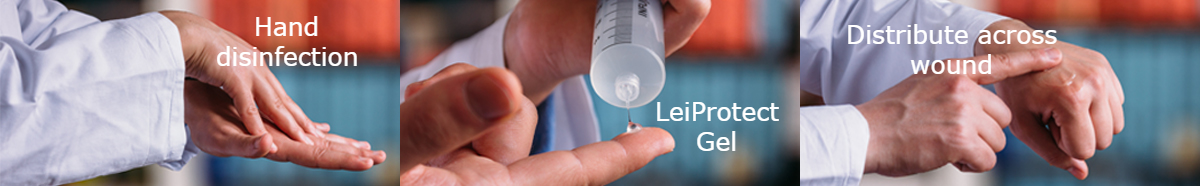
LeiClean beinhaltet, dass sich die Anwender des Film-bildenden Gels LeiProtect®, einem Medizinprodukt der Klasse IIb, zunächst wie von der WHO empfohlen ihre Hände mit hydroalkoholischer Lösung desinfizieren, ehe sie sich ihre Hautwunden mit dem filmbildenden Gel LeiProtect® abdecken.
LeiProtect® haben die Apotheker und Ärzte unseres Waisenmedizin e. V. in den Anfängen 2014 in Afghanistan entwickelt.
Das sonderzugelassene Gel LeiProtect® Gel stellt die GMP-zertifizierte Krankenhausapotheke der Universität Erlangen als OEM-Hersteller für den Waisenmedizin e.V. als verantwortlichen Hersteller her. Die Kosten der mit einem Jetstreamer der Fa. YSTRAL nach GMP hergestellten kleinen 10 x 1 kg Chargen von LeiProtect® sind bisher noch relativ hoch. Sie betragen derzeit noch 1 € pro 1 g Gel, was derzeit noch zu täglichen Behandlungskosten von 0,1 bis 0,2 € pro Hautläsion führt, die bei der Herstellung von 1.000 kg Chargen erheblich, d.h. um ca. 90% sinken dürften. Der Conti-TDS Jetstreamer ist eine Materialspende im Neuanschaffungswert von 70.000 € an den gemeinnützigen Waisenmedizin e. V..
Unsere vorläufigen Ergebnisse einer „compassionate use“ Beobachtung an 35 Patienten mit 45 komplexen Hautleishmaniose Läsionen von algerischen Patienten, die sich für eine LeiClean-Behandlung entschieden hatten, weil sich nicht einer für solche Fälle vorgesehenen zwei Wochen langen Krankenhausbehandlung mit i. m. Spritzen von fünfwertigen Antimon unterziehen wollten, zeigten, dass auch ohne Antimon Chemotherapie ein ambulante Heilung der Leishmania Hautläsionen erreicht werden konnte. Die Hälfte der Patienten zeigte eine Heilung ihrer komplexen Leishmania Läsionen nach 4 Wochen mit durchschnittlich 7 € pro Läsion für die LeiProtect®-Kosten zum derzeitigen tatsächlichen Produktionspreis. Im Vergleich dazu: Die Kosten einer dreiwöchigen Krankenhausbehandlung mit fünfwertigem Antimon wurden in einer kürzlich in Marokko erschienenen Publikation untersucht (Tachfouti N, Najdi A, Alonso S, Sicuri E, Laamrani El Idrissi A, Nejjari C, Picado A). Kosten für die Behandlung der pädiatrischen viszeralen Leishmaniose in Marokko. PLoS One. 2016 Jun 3;11(6):e0155482). Die stationären Behandlungskosten wurden auf USD 510 pro Patienten geschätzt.